Published: A.V. Kurguzova, S.Z. Smirnov, Yu.I.Klyukin, N.S. Karmanov Inclusions of Bi_Rich Solutions in Quartz from Zwitter Related to Li–F Granite in the Severny Pluton, Chukchi Peninsula: A View on Bismuth Behavior in Process of Greisenization// Geology of Ore Deposits. 2014.Vol. 56. №8. – pp. 1-8
Abstract—General characteristics of fluids, which were responsible for formation of zwitters within Severny
massif, were constrained on the basis of fluid inclusion research of zwitter quartz. Fluid inclusions, which
contain Bi oxyhalogenide crystalline phases, are discussed in details. These data provide insights into the spe
ciation of bismuth in the course of its transport by postmagmatic fluids related to Li–F granites.
DOI: 10.1134/S1075701514080030
INTRODUCTION
This paper represents results of fluid inclusion study of quartz from fine_grained greisen (zwitter)
genetically related to Li–F granite of the Severny (Northern) pluton in the Chaun district, Chukchi Peninsula are reported. In particular, we report new information on revealed Bi oxychloride compounds in fluid inclusions. These results are noteworthy, because data on Bi concentrations in natural hydrothermal fluids and its speciation remains limited.
GEOLOGY OF THE SEVERNY PLUTON
The Severny pluton is situated on the eastern coast of the Chaun Bay and extends in a northwestern direction coinciding with the strike of the country Upper Triassic metamorphosed sandshale country rocks of the Palyavaam Megasynclinorium. Judging by the remnants of hornfels in roof, the erosion level is not deep. The pluton is primarily composed of porphyritic biotite granite, protolithionite granite, granite porphyry, pegmatite, and aplite veins (Marin and Alekseev, 1992). Microcline–albite granite and ongonite with zinnwaldite and topaz are the youngest igneous rocks (Alekseev et al., 2013).
Granitic rocks of the Severny pluton are greisenized. The dark_mica greisens (zwitters) described from Li–F granites in the Erzgebirge, Mongolia, and Transbaikalia (Kovalenko, 1974; Štemprok, 2005; Borodkin, 2012) are also developed in the Severny pluton, where they occurs as irregular pockets and narrow lenses a few decimeters in thickness. Two types of zwitters are recognized: (i) melanocratic fine_ to medium_grained massive mica varieties and (ii) leucocratic quartz and quartz–topaz varieties; both were the objects of our study (Figs. 1a, 1b).
ACCESSORY MINERALIZATION OF ZWITTER
Zwitters in the Severny pluton are characterized by a combination of lithophile and chalcophile mineralization. The following accessory minerals have been identified: Th_ monazite, wolframite, wolframoixiolite, W–Nb rutile, columbite, As_thorite, as well as loellingite, pyrite, chalcopyrite, bismuthinite, native bismuth, and rooseveltite. Arsenic and bismuth closely associate in zwitter mineralization due to their geochemical affinity (Fig. 1c). Bi and As minerals are spatially associated with Th_zircon (Fig. 1d).
METHODS
Doubly polished thin sections of rocks and hand picked quartz grain separates (_1.0+0.5 mm) were used for the fluid inclusion study. The phase composition of inclusions was characterized, and solid phases were identified using Raman spectroscopy (we employed a Horiba Labram HR 800 liquid_nitrogen cooled CCD detector with a Horiba Scientific Symphony II and a 514 nm_laser at the Institute of Geology and Mineralogy, Siberian Branch, Russian Academy of Sciences in Novosibirsk). Quartz grains containing fluid inclusions were separated under optical microscope in immersion liquid (glicerol). After separation grains with inclusions were crushed in jasper mortar, and the fragments were mounted on a metallic pellet for subsequent SEM study.

The dry salt residues left in inclusions after opening were analyzed on SEM equipped with a source of electrons and EDS (Oxford Instruments). The accelerating voltage of 20 kV and the beam current of about 1 nA were applied for compositional analysis. The electron beam was ~10 nm in diameter. Many phases identified with this technique appeared to be instable under an electron beam. To minimize the effect of this instability, analysis was performed by scanning over an area of 10 ×10 to 100 × 100 μm depending on the dimensions of the objects. The morphology of the phases and their orientations relative to the detector did not allow us to use the full capability of the instrument, which was intended for quantitative analysis.
Therefore the semiquantitative ratios of components were recalculated to atomic ratios. Only nearly horizontally oriented areas of grains not screened by walls of opened inclusions or other phases were used.
FLUID INCLUSIONS IN QUARTZ FROM ZWITTERS
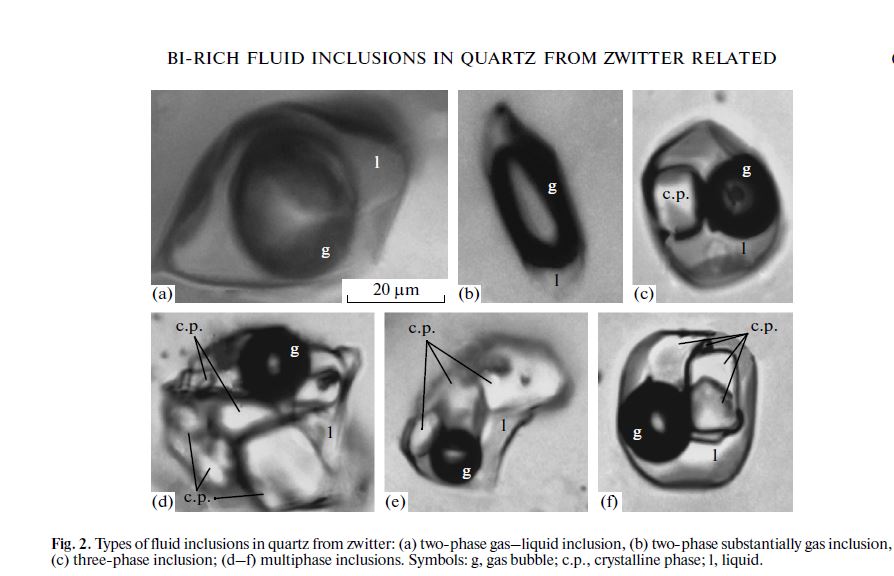
Fluid inclusions in zwitter have different phase compositions. Two_phase gas_rich inclusions (Figs. 2a, 2b), as well as multiphase crystal_rich inclusions are observed. Our main attention was focused on multiphase inclusions (Figs. 2d–2f). Based on Raman spectra of the daughter minerals in the fluid inclusions, we assume that one of the crystalline phase is Fe chloride. Neither bismuth compounds nor native bismuths were identified with Raman spectroscopy.
The crystalline phases were revealed as products of evaporation of solution from opened vacuoles and on the immediately adjoining quartz surface. Two hundred thirty opened inclusions have been examined. The frequencies of elements identified in opened inclusions are summarized in Table 1. Fe, Na, and K, as well as Mn and Zn chlorides dominate in the dry residue. The opened inclusions are showed in Fig. 3.
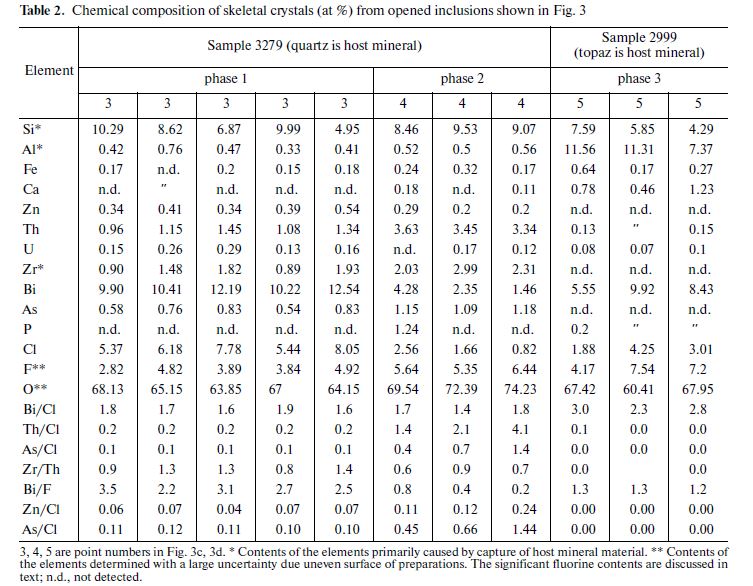
In addition, inclusions with unusual Bi oxychlorides are predominant in dry residue (Figs. 3c, 3d). The chemical compositions of the crystals indicated by numerals in Figs. 3c and 3d are given in Table 2. It is noteworthy that crystals of oxychlorides in the dry residues have skeletal shapes (Fig. 3c).
Inclusions with bismuth oxychlorides in dry residue are less abundant than inclusions with alkali metal chlorides and are spatially related to crystalline inclusions of zircon and thorianite in quartz. The main components of dry residues of the inclusions with high Bi concentrations are represented by O, Bi, Cl, and Th, while As, U, Ca, Zn, and Fe occur in subordinate amounts. In contrast to most fluid inclusions, K, Na, and Ca chlorides were not identified as crystalline phases of dry residues of Bi_rich ones.
Since geometry of dry residues is not suitable for performing good EDS analyses, only compositions with a total percentage of 100 } 20 wt % had been selected for further interpretation. The compositions were then compared on the basis of their atomic concentrations. Si and Al concentrations largely dependent on contamination by host minerals were not taken into account. Fluorine contents in dry residues of inclusions from sample 3279 (quartz is a host mineral) were regarded as a rough estimate. These fluorine concentrations vary from 1 to 4 wt % with a high uncertainty (~±0.3 wt % at 1σ). Based on atomic proportions, three compositions conditionally called phases 1, 2, and 3 in Table 2 can be selected from among the dry residues suitable for interpretation.
Phase 1 is Bi oxychloride with atomic Bi/Cl ratio ranging from 1.6 to 1.9; with lower Th, As and higher U, Zn contents. Phase 2 is characterized by lower Bi and Cl contents at the same Bi/Cl ratio (1.4–1.8) as in phase 1. However, this phase is distinguished by the highest Th and As contents. The U and Zn concentrations in phase 2 are somewhat lower than in phase 1; Ca has been detected in small amounts. Elevated P contents are detected at some points. Higher F concentrations are systematically detected in phase 2. The composition corresponding to phase 3 has been established in the dry residue of an inclusion hosted in topaz. This phase differs from others in its somewhat lower Bi and Cl contents at a higher Bi/Cl ratio (2.3–3.0). This phase is also characterized by the highest Ca content. The analyses yield somewhat higher F content relative to phases 1 and 2. Phase 3 is distinguished by the lowest Zn, Th, U, and As concentrations.
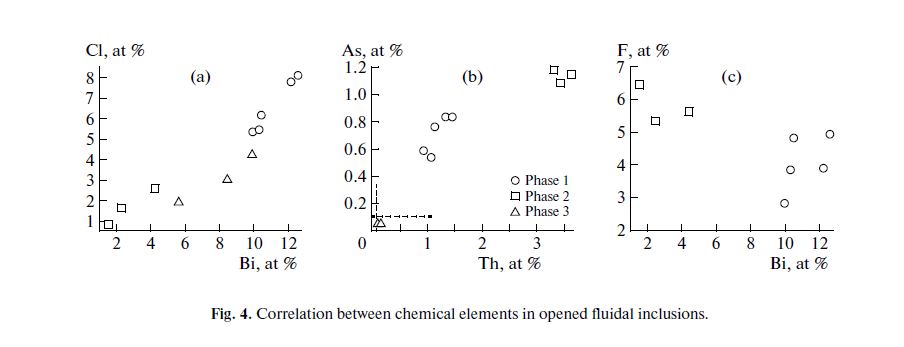
The correlations between Bi, Cl, F, As, and Th in dry residues of the inclusions in sample 3279 are shown in Fig. 4. The positive correlation between Bi and Cl is evident for all three phases (Fig. 4a). The correlations between Th and As (Fig. 4b) and between Bi and F (Fig. 4c) are not so distinct within each phase. At the same time, Th and As reveal a positive correlation, whereas Bi and F are characterized by negative correlation.
Bismuth and chlorine are not contained in host minerals, so that these elements must have originated from the opened fluid inclusions. Thorium is incorporated into zircon, which occurs as a crystalline inclusion in quartz near the opened fluid inclusion (Fig. 3c).
Table 2 shows that Zr and Th are also characterized by a positive correlation. According to the EDS results, Th occurs as an impurity in zircon and the atomic Zr/Th ratio = 8.3. In the high_Th phase 2, this ratio is 0.6–0.9, whereas in phase 1, with a lower Th content, it is 0.8–1.4. Thus, the contribution of crystalline zircon inclusion to the composition of phases 1 and 2 in dry residue is evident, however, the much lower (down to inverse) proportions of Zr and Th show that the greater part of Th in phase 2 was derived from the substance of fluid inclusion. Phase 3, in the dry residue of the inclusion in topaz, is enriched in fluorine, and this may be a result of contamination with material of the host mineral. At the same time, lower but comparable fluorine concentrations are also contained in phases 1 and 2. This implies that fluorine is also a component of the entrapped fluid. The available data are insufficient for assessing the contribution of fluorine from the composition of entrapped fluid because of the high uncertainty of determination.
The above data show that the phases detected in opened inclusions are products of the crystallization of the solutions enriched in Bi, Cl, Th, As, and F. Bismuth oxychloride likely is one of dominating phases.
Judging by the Bi/Cl ratio it can be referred to the bismoclite BiClO–daubreeite BiO(Cl, OH) series (phase 1).
The Th–As compound is the second in significance (phase 2). The composition of phase 3 assumes that Bi oxides and hydroxides can also be components of dry residues. In addition to Bi, Cl, Th, and As, Fe, Zn, U, and Ca occur as admixtures in dry residues of fluid inclusions. Most likely they compose easily soluble chlorides (Fe, Zn, or Ca) or oxygen compounds (U).
The compositions of dry residues show that the aqueous solutions responsible for the formation of zwitters were variable in composition. The first type of solutions was represented by Na and K chloride fluids with Ca and Fe admixtures. The second type was not known earlier. Judging from the compositions of the dry residues, these are chloride or fluorine–arsenic–chlorine solutions of Bi, Th, and U with addition ofFe, Zn, and Ca admixtures.
Bismuth oxyhalogenides are rare natural substances with such anions as OCl (bismoclite, daubreeite), OF (zavaritskite), FCl (zhangpeishantite), and some others. Bismoclite is a mineral of a tetragonal system with a layered structure (Palache et al., 1951; Keramidas et al., 1993). Typically, bismoclite occurs as a secondary mineral in granite pegmatite and grease and was described for the first time in alluvium along side granite pegmatite (Frondel, 1943). Bismoclite is also known as a secondary mineral in Au–Bi quartz–adularia hydrothermal veins in the Punta del Fenaio monzogranite, Italy (Dini and Orlandi, 2010). At the pitchblende deposits near Dalbeattie in South Scotland, bismoclite makes up aggregates of yellow_green secondary grains in association with mixite, bismutite, walpurgite, and eulytite (Braithwaite and Knight, 1990). Zavaritskite was described in greisen and veins at the Sherlova Gora deposit as a secondary mineral after bismuthinite (Dolomanova, 1962).
BISMUTH AND ARSENIC IN POSTMAGMATIC PROCESSES
Arsenic, antimony, and bismuth are elements from the 15th group of periodic periodic table of the elements; they belong to semimetals and have a number of similar geochemical properties. They occur in minerals as anions and cations, reveal an inclination to the formation of intermetallic compounds, and occur in a native state. Oxygenic and halogenic compounds of these elements are extremely rare. Bismuth reveals an affinity to As, Sb, and Pb and replaces these elements in minerals from greisen. In most publications concerned with As–Bi fluids, the behavior of these elements is regarded in context of the formation of gold deposits (Skirrow, 2002; Tooth, 2008).
It was shown in previous publications on behavior of As and Sb that hydroxyl species of these elements are predominant even at significant Cl contents in aqueous solutions (Pokrovski et al., 1996, 1999).

Under oxidizing conditions, the medium_ to high temperature acidic to neutral aqueous fluids transfer
As3+ mainly in the form of As(OH)3 (Pokrovski et al., 2002). Arsenopyrite is the major As mineral in greisens mineral assemblages. The formation of arsenopyrite is related to the change of conditions from oxidizing to reducing along with a temperature drop according to the following reaction (Heinrich, 1986):

FeCl2(aq) + As(OH)3(aq) + H2S(aq) + H2(aq)= FeAsS + 3H2O + 2H+ + 2Cl–.
Finding of arsenolite in fluid inclusions in quartz from the Muiane pegmatite (Banks et al., 2011) is additional evidence for high As contents in fluids related to the crystallization of granitic magma and its transfer with fluid in the oxidized state. High As concentrations were established in fluid inclusions hosted in miarolic quartz of the Leskhovskaya pegmatite vein in the southwestern Pamirs (Smirnov et al., 2011). Information on Bi content and behavior in hydrothermal systems remains limited. As has been shown in a few publications (Kolonin and Laptev, 1982; Wood et al., 1987), Bi3+ is the main valence state of this element in hydrothermal solutions at a temperature below 300°C. Bi5+ is a strong oxidant in aqueous solutions (Cotton et al., 1999). In addition, a stable solution with Bi+ has been obtained by dissolving native Bi in a concentrated BiCl3(s)–HCl solution at room temperature. It turns out, however, that the univalent state is instable when heated to 100°C (Ulvenlund and Bengtsson, 1994, 1995). The most popular model (Kolonin and Laptev, 1982; Pokrovski, 2002; Kruszewski, 2009) suggests a Bi transfer by aqueous fluid as hydroxyl species BiO3 and Bi(OH)3. Bismuth is a strong Lewis acid and is subjected to hydrolysis at pH 1. In a highly acidic medium (pH 1–5), Bi6O4 complexes occur, whereas Bi(OH)3 is stable under nearly neutral conditions.
Thus, bismuth, as well as arsenic and antimony, is largely transferred as neutral hydroxyl species within a wide range of temperature, pressure, and fluid regimes. However, in contrast to As and Sb, the Bi hydroxyl species are less soluble in hydrothermal fluids (Tooth et al., 2013) and require a temperature above 400°C for their transfer. These data are consistent with occurrence of Bi minerals in the high_temperature mineral assemblages.
The study of other possible (chloride, sulfide) species of dissolved bismuth remains an important task. Wood et al. (1987) indicated that at 200–350°C bismuth most likely occurs in aqueous solutions as hydroxyl species, but in chlorine_bearing systems its transport as a chloride complex is possible. Tooth et al. (2013) suggested that Bi chloride complexes dominate in high_saline solutions (4m Cl solution) especially at a temperature exceeding 300°C.
DISCUSSION
The discovery of Bi oxychlorides close to bismoclite or daubreeite in composition in dry residues of opened fluid inclusions shows that the Bi concentration in fluid inclusions is rather high. At the same time, no Bi minerals as daughter phases have been observed in fluid inclusions at room temperature. The skeletal shape of crystals and their arrangement relative to the open vacuole serve as evidence for their formation after opening of inclusions. Bismuth chloride BiCl3
can be readily produced by direct interaction of the molecular or ionic chlorine with bismuth. In oxidized solutions bismuth chloride can occur as a product of interaction of Bi oxide with HCl. However, in the presence of water, Bi chloride becomes unstable and is according to the reaction
Bi3+ + 3Cl– + H2O → BiOCl↓ + 2HCl.
This reaction is suppressed by acidation with HCl. Bi hydroxide, which is easily dissolved in hydrothermal solutions, is practically insoluble in water under normal conditions(1.4 × 10–4 g/100 mL water). As a result, it can be suggested that opened inclusions initially contained an aqueous solution that had an acidic reaction due to excess HCl or chlorine ions, which were not compensated by alkali metals. Bismuth occurred in such solutions in chloride or ionic form. It can be suggested that the opening of the inclusion and the accompanying pressure release resulted in the removal of excess HCl in a gaseous phase, and hydrolysis of bismuth chloride gave rise to the formation of bismoclite or daubreeite.
Thus, the Bi oxychloride detected in dry residues of fluid inclusions in quartz from zwitters of the Severny pluton can serve as evidence for chloride complexing of Bi dissolution and transfer in postmagmatic acid fluids. The rarity of such fluid inclusions shows that fluids with high Bi concentrations mark only a short episode of mineral formation. It is noteworthy Bi that oxychloride has only been detected in dry residues in the fluid inclusions, which are in contact with zircon and thorite crystals. (Fig. 5). Taking into account high Th and As concentrations in the dry residue (Table 1), it may be concluded that the aqueous solutions with high Bi content also transferred Th and As. This implies that the fluids represented by inclusions with high Bi, Th, and As contents controlled the formation of accessory As and Bi minerals (rooseveltite, chernovite, atelestite), zircon, and thorite in the course of greisenization of the Severny pluton.

CONCLUSIONS
Minerals close in composition to bismoclite and daubreeite were identified in the dry residues of certain fluid inclusions in quartz from zwitters of the Severny pluton. The occurrence of these minerals assumes that fluids themselves were oxidized and acidic and that Bi was dissolved in fluid as chloride and partly fluorine species.
In addition to bismuth, the fluids contained much Th and As, and this facilitated the formation of accessory arsenopyrite, atelestite, native bismuth, rooseveltite, thorite, and zircon in zwitters.
ACKNOWLEDGMENTS
This study was supported by the Ministry of Education and Science of the Russian Federation, state contract no. 5.2115.2014/K.
REFERENCES
Alekseev, V.I., Marin, Yu.B., Kapitonov, I.V., and Sergeev, A.S., Age and material sources of lithium_fluorine granites of the Far East (U–Pb and Lu–Hf isotope data), Dokl. Earth Sci., 2013, vol. 449, no. 2, pp. 444–446.
Banks, D., Bodnar, R., and Bozkaya, G., Exotic pegmatite fluids: A precursor for orogenic gold mineralization, Berichte der Geologische Bundesanstalt, 2011, vol. 87, p. 213.
Borodkin, N.A. and Pristavko, V.A., Identification of zwitter from petrochemical and geochemical criteria, Otech. Geol., 2012, no. 4, pp. 49–56.
Braithwaite, R.S.W. and Knight, J.R., Rare minerals, including several new to Britain, in supergene alteration of U–Cu–As–Bi–Co mineralisation near Dalbeattie, South Scotland, Mineral. Mag., 1990, vol. 54, pp. 129–131.
Ciobanu, C.L., Cook, N.J., and Pring, A., Bismuth tellurides as gold scavengers, in Mineral Deposit Research: Meeting the Global Challenge, Berlin: Springer, 2005, pp. 1387–1390.
Dini, A. and Orlandi, P., A new gold–bismuth occurrence at Punta del Fenaio (Giglioisland, Tuscany), Mem. Atti della Societa Toscana di Scienze Naturali, 2010, vol. 115, pp. 73–82.
Frondel, C., New data on agricolite, bismoclite, koechlinite, and the bismuth arsenates, Am. Mineral., 1943, vol. 28,
pp. 536–540.
Heinrich, C.A. and Eadington, P.J., Thermodynamic predictions of the hydrothermal chemistry of arsenic, and theirsignificance for the paragenetic sequence of some cassiterite–arsenopyrite–base metal sulfide deposits, Econ. Geol., 1986, vol. 81, pp. 551–529.
Keramidas, K.G., Voutsas, G.P., and Rentzeperis, P.I., The crystal structure of BiOCl, Zeitschr. Krist., 1993, vol. 205, pp. 35–40.
Kolonin, G.R. and Laptev, Y.V., Study of process of dissolution of a_Bi2O3 (bismite) and complex formation of bismuth in hydrothermal solutions, Geokhimiya, 1982, vol. 11, pp. 1621–1631.
Kovalenko, V.I., Kuz’mina, M.I., Gundsambuu, Ts., et al.,
Geochemical characterization of a new_type zwitter as Sn bearing metasomatic rock in Mongolia (composition, zoning, and parageneses), Dokl. Akad. Nauk SSSR, 1970, vol. 190, no. 3, pp. 690–693.
Kovalenko, V.I., Kuz’min, M.I., and Kozlov, V.D., Metasomatic twitters and related rare_metal mineralization: A case of deposits in Mongolia and Czechoslovakia, in Metasomatizm i rudoobrazovanie (Metasomatism and Ore Formation), Moscow: Nauka, 1974, pp. 42–53.
Kruszewski, J.M. and Wood, S.A., Experimental measurement of the solubility of bismuth phases in water vapor from 220°C to 300°C: Implications for ore formation, Appl.
Geochem., 2009, vol. 24, pp. 493–503.
Marin, Yu.B. and Alekseev, V.I., Razrabotka kriteriev lokal’nogo prognozirovaniya olovyannogo orudeneniya namassive Severnom na osnove izucheniya metasomaticheskoi mineralogicheskoi i geokhimicheskoi zonal’nosti (Development of Criteria of Local Forecasting of Tin Mineralization in the Severny Pluton on the Basis of Studying Metasomatic, Mineralogical, and Geochemical Zoning), St. Petersburg:
St. Petersburg Mining Inst., 1992.
Palache, C., Berman, H., and Frondel, C., The System of Mineralogy of James Dwight Dana and Edward Salisbury
Dana, 1951, p. 1124.
Pokrovski, G., et al., Thermodynamic properties and stoichiometry of As(III) hydroxide complexes at hydrothermal conditions, Geoch. Cosmochim. Acta, 1996, vol. 60,
pp. 737–749.
Pokrovski, G.S., Berny, J._M., and Zotov, A.V., Solubility and Raman spectroscopic study of As(III) speciation in organic compound–water solutions: A hydration approach for aqueous arsenic in complex solutions, J. Sol. Chem., 1999, vol. 28, pp. 1307–1327.
Pokrovski, G., Kara, S., and Roux, J., Stability and solubil_
ity of arsenopyrite, FeAsS, in crustal fluids, Geochim. Cosmochim. Acta, 2002, vol. 66, pp. 2361–2378.
Shin, D., Park, H._I., Lee, I., et al., Hydrothermal As_Bi mineralization in the Nakdong deposits, South Korea:
Insight from fluid inclusions and stable isotopes, Can. Mineral., 2004, vol. 42, pp. 1465–1481.
Skirrow, R.G. and Walshe, J.L., Reduced and oxidized Au Cu–Bi iron oxide deposits of the Tenant Creek Inlier, Australia: An integrated geologic and chemical model, Econ.Geol., 2002, vol. 97, pp. 1167–1202.
Smirnov, S.Z., Astrelina, E.I., Ragozin, A.L., et al., Latemagmatic crystallization of tourmaline_bearing granite pegmatites: A case of the Shakhdara and Leskhoz veins, the southwestern Pamirs in Granity i protsessy rudoobrazovaniya. Materialy konferentsii, posvyashchennoi pamyat akademika V.I. Kovalenko (Granites and Ore Formation. Proceedings of Conference in Memory of Academician V.I. Kovalenko), Moscow, 2011, pp. 122–123.
Stemprok, M., Pivec, E., and Langrov, A., The petrogenesis of a wolframite_bearing greisen in the Vykmanov granite stock, Western Krušnйhory pluton (Czech Republic), Bull.
Geosci., 2005, vol. 80, no. 3, pp. 163–184.
Tooth, B., Brugger, J., Ciobanu, C., et al., Modeling of gold scavenging by bismuth melts coexisting with hydrothermal fluids, Geology, 2008, vol. 36, pp. 815–818.
Tooth, B., Etshmann, B., Pokrovski, G.S., et al., Bismuth speciation in hydrothermal fluids: An X_ray absorption
spectroscopy and solubility study, Geochim. Cosmochim.
Acta, 2013, vol. 101, pp. 156–172.
Wood, S.A., Crerar, D.A., and Borcsik, M.P., Solubility of the assemblage pyrite–pyrrhotite–magnetite–sphalerite–
galena–gold–stibnite–bismuthinite–argentite–molybdenite in H2O–NaCl–CO2 solutions from 200 to 350°C,
Econ. Geol., 1987, vol. 82, pp. 1864–1887.
Translated by V. Popov